
*Repeat until the most accurate data is collected. Estimate the freezing point of t-butanol from the data collected.Using a temperature probe connected to the Sparkvue software create a graph of.Melt the t-butanol in a test tube using a warm water bath.Part I – Determination of Kf of T-Butanol In this lab, we are going to determine the freezing point depression of t-butanol, and toĬalculate the molar mass of an unknown substance using the relationship between ΔTf and Which characterizes the solvent used, Kf. To calculate the extent of the depression using the molality of the solution, and the constant When a solid is dissolved in a liquid, the freezing point of that liquid is depressed. Pressure lowering, boiling point elevation, freezing point depression, and osmotic pressure. Molecules or ions, but not upon the identity of the solute. The Methodology of the Social Sciences (Max Weber)ĭetermination of Molar Mass by Colligative Properties A freezing Point Depression Experiment Name: Shaili Batsri Date: 09/07/ Lab Partners: Chris Zalayes Introduction:Ĭolligative properties of solutions are properties that depend upon the concentration of solute.Bursten Catherine Murphy Patrick Woodward) 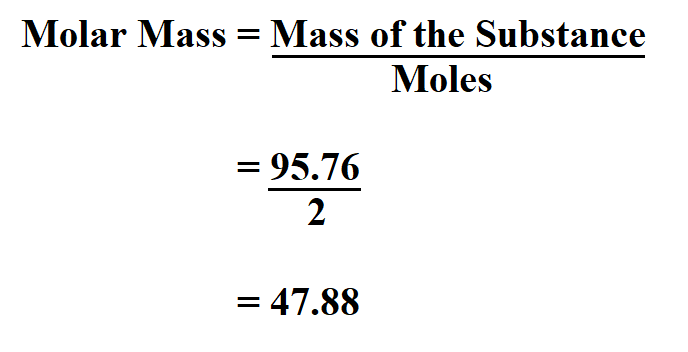
0 Comments
Leave a Reply. |


 RSS Feed
RSS Feed
